Kenya Includes Contraceptive Injections in Its Pharmacist Training Package
An Advocacy Journey

Working alongside family planning advocates, Jhpiego Kenya applied the nine-step SMART advocacy approach to engage stakeholders in the creation of a new pharmacist training package. The updated curriculum includes instruction on providing contraceptive injectables DMPA-IM and DMPA-SC.
On July 13, 2020, Kenya’s Department of Family Health approved a new national training package for pharmacists and pharmaceutical technologists that includes subcutaneous and intramuscular DMPA (DMPA-SC and DMPA-IM). The comprehensive curriculum integrates family planning, HIV, sexually transmitted infections, and other related services.

According to a 2014 Kenya Demographic and Health Survey, one in 10 family planning clients obtained contraceptives from pharmacies. However, it is worth noting that prior to 2018, pharmacists and pharmaceutical technologists could only provide condoms and pills. Pharmacists needed to refer clients who opted for injectable contraceptives to other providers, despite WHO guidelines recommending that pharmacists could do so safely and effectively when trained.
Advance Family Planning (AFP) local partner Jhpiego Kenya advocated alongside like-minded organizations for more than three years. They engaged decision-makers to support the policy change. This write-up provides details of the advocacy journey, including the following aspects:
- Advocacy approach.
- Policy change.
- Development of pharmacist training package.
It also outlines next steps in the national scale up of the pharmacists’ training.
Advocacy Approach
Working with family planning advocates, Jhpiego Kenya applied the nine-step SMART advocacy approach in engaging with decision-makers.
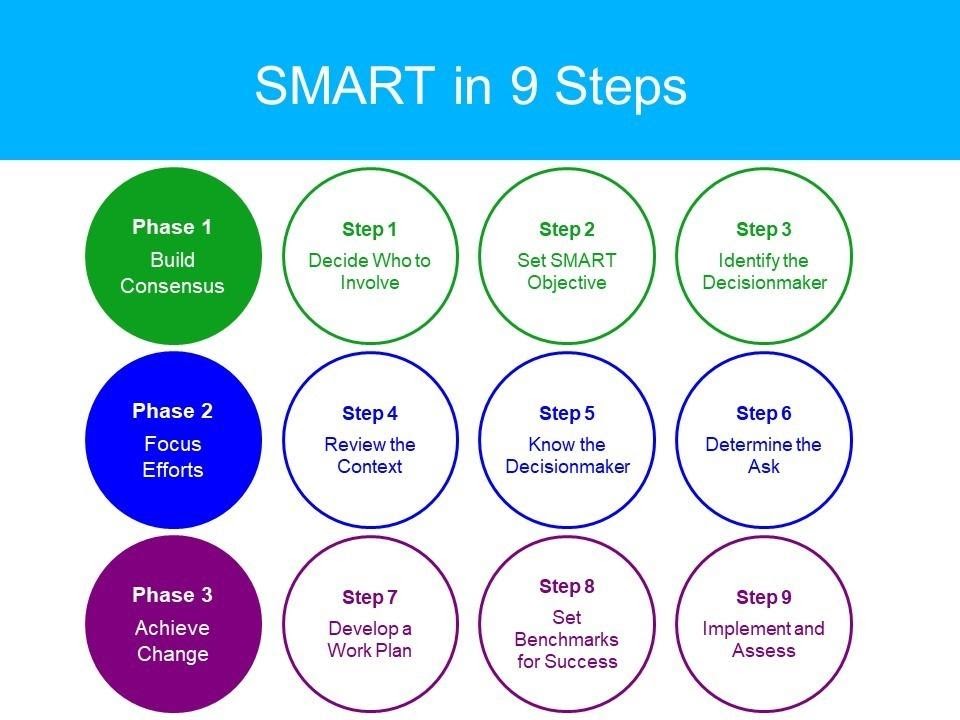
The approach also incorporates a landscape assessment that provides essential contextual and strategic information that informs the advocacy efforts. Following the landscape assessment, advocates identify critical champions or stakeholders to be involved in advancing the advocacy efforts. The next step entails developing an objective that is SMART—Specific, Measurable, Attainable, Relevant, and Time-bound. Advocates will then identify and analyze the decision-maker; this is the person who has the power to ensure that the advocacy issue is addressed. They will determine the ASK—what is it that they want the decision-maker to do. This will include creating messages that argue the case. Advocates will also identify the messenger—a person who is listened to by the decision-maker.
Upon completion of the steps, advocates will develop an advocacy workplan, present their case, monitor the implementation, and document the results. On October 13, 2021, Advance Family Planning launched an updated SMART approach that reorganized and renamed some of the steps while retaining the basic structure. The experiences of advocates in developing and implementing the SMART approach informed the adjustments of the new look:
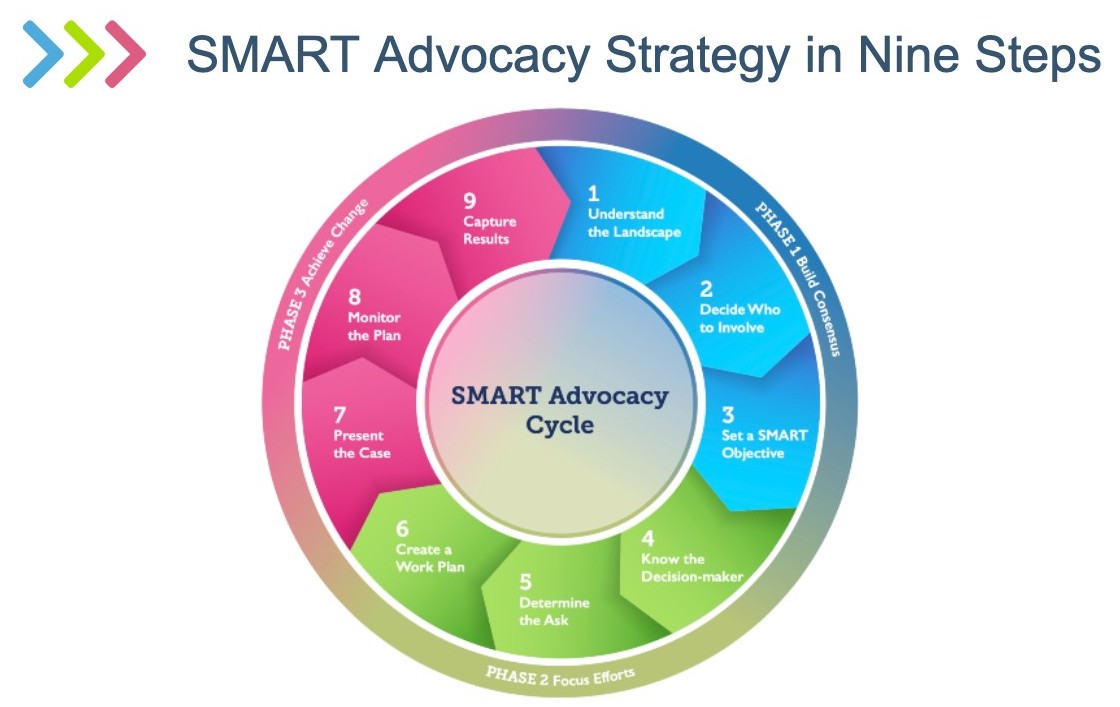
The updated SMART Advocacy approach includes a user’s guide that helps advocates who want to plan and execute an advocacy strategy session. It is available in English, French, and Spanish.
Implementing the SMART Approach to Achieve Policy Change
On October 9, 2018, Kenya’s Ministry of Health (MOH) amended its family planning guidelines to allow pharmacists and pharmaceutical technologists throughout the country to provide contraceptive injectables (DMPA-IM and DMPA-SC). This policy change opened up an alternative for women and young people who may be reluctant to seek contraception at health facilities. It also expanded the role of the private sector in helping reduce commodity shortfalls.
The advocacy journey started with conducting the landscape assessment that clearly mapped out the environment, actors, evidence, and the policy context. The nine-step SMART approach guided the advocacy strategy development.
Early in the strategy implementation, advocates held a meeting with the president and the chief executive officer of the Pharmaceutical Society of Kenya (PSK), a professional body for pharmacists in Kenya. The meeting sought to enlist their support for a policy change that would allow pharmacists to provide injectable contraceptives. Following this meeting, the president and CEO successfully advocated for the policy change before the full PSK Council. Given that pharmacists were responsible for implementation of the policy change, buy-in from the professional body was critical.
Jhpiego Kenya then facilitated a joint meeting between the MOH and the PSK to explore the policy change. Both parties agreed that only pharmacists trained in administering injectable contraceptives would provide the method. The pharmacy outlets would also require a counselling area and a commitment to submit data to the MOH.
Advocates suffered a major setback, however, when the leadership of both MOH and PSK transitioned. This meant going back to the drawing board to engage with the new leadership and earn their commitment. Once this was achieved, the Ministry of Health (with support from Jhpiego and the Clinton Health Access Initiative), convened a stakeholders’ meeting in June 2018 to review and finalize the National Family Planning Guidelines for Service Providers, 6th Edition.
Following the advocacy efforts for the policy change, the MOH agreed to amend the guideline. The amendments were approved via national validation.
Development of Training Package
Before they could offer injectable contraceptives, pharmacists and pharmaceutical technologists needed in-service training on how to administer them properly and safely. Amending a policy to allow pharmacists and pharmaceutical technologists to provide the methods did not necessarily mean that the policy will be implemented. In April 2019, Jhpiego Kenya led advocacy efforts to engage the MOH on next steps.
On April 24, 2019, the family planning manager at the MOH formed a team to plan the implementation of the new policy provision, including the development of a training package. The team comprised officers drawn from national and subnational governments, regulatory agencies, professional bodies, supply chain actors, and private sector and implementing partners. It included participants from the several organizations.
MOH Implementation Team (click to expand)
- Ministry of Health
- Pharmacy and Poisons Board
- Pharmaceutical Society of Kenya
- Kenya Pharmaceutical Association
- Kenya Medical Supplies Agency
- County Pharmacists
- John Snow, Inc. (Access Collaborative)
- Clinton Health Access Initiative
- UNFPA
- Population Services Kenya
- Marie Stopes Kenya
- Health Strat
- DKT
- Bayer
- Pfizer
- Jhpiego
Following the completion of the implementation plan in April 2019, the team embarked on the pharmacist training package. The package (or “curriculum”) comprises a trainer’s manual, participant manual, and a participant logbook, in which a trainee logs all procedures for certification.
Core competencies covered in the package include:

- Counseling for family planning.
- Medical eligibility criteria.
- Service integration (HIV and related service) and referrals.
- Contraceptive methods.
- Pharmacovigilance.
- Infection prevention and control.
- Anatomy and physiology of the human reproductive system.
- Reproductive health commodity management.
- Family planning documentation and reporting.
The package clearly outlines issues of accreditation for pharmacy outlets and the certification of trainees. Once trained, pharmacists will be able to orient clients on self-injection of DMPA-SC as part of an implementation science initiative.
The training package was pretested with a group of 15 pharmacists selected as a national trainer of trainers effort. The pretest was completed on August 16, 2020, and validated on December 20, 2020, at meetings convened by the MOH. The experience and inputs from the two meetings were used to refine the training package.
Once a training package is validated, the next step is the official MOH sign-off. Unfortunately, this did not happen as anticipated. The MOH program manager leading the training package development and the head of the Department of Family Health transitioned. Their replacements needed to be brought on board.
Nearly six months passed with little progress. On June 23, 2020, AFP partner Jhpiego Kenya mobilized a few champions within and outside MOH to provide a briefing to the new head of the Department of Family Health and make a case for signoff. The champions included:
- Former MOH family planning managers who had worked on the training package.
- CEO of the PSK (who is also PATH’s Access Collaborative Ambassador).
- Director of Training at the Pharmacy and Poisons Board.
- CEO of the Urban Research and Development Centre for Africa.
The head of MOH’s Division of Reproductive and Maternal Health Services also attended the meeting.
Following this meeting, the head of the Department of Family Health asked Jhpiego Kenya to provide him with hard-copy documents that confirmed prior MOH approval for pharmacists to provide injectable contraceptives. Jhpiego was also asked to respond to how quality standards will be addressed.
Jhpiego Kenya held a meeting with the head of the Department of Family Health on July 2, 2020, to provide the necessary information. On July 13, 2020, the Departmental Head signed off the training package.
Following this milestone, Jhpiego Kenya engaged with the new MOH family planning program manager to ask for the establishment of a training coordination committee. It was established on August 13, 2020. The family planning training coordination committee for pharmacists and pharmaceutical technologists is co-chaired by the Department of Family Health and the Pharmacy and Poisons Board. The PSK which has been a key ally in the development of the training package, serves as the secretariat.
Lessons Learned (click to expand)
Identifying priority advocacy objectives
Advocacy objectives and wins are incremental. An enabling policy environment is an essential outcome prior to advocating for a training package or curriculum for pharmacists and pharmaceutical technologists.
Role of messenger and allies
Engage the key technical officer within the MOH as a messenger—a person listened to by the decision-maker. Make the pharmaceutical professional bodies your key advocacy allies prior to engaging with the MOH. You cannot ask the MOH to make a policy change or develop training materials when the implementers of your advocacy “ask” are not in sync with you.
Role of champions
Even when decision-makers transition, you can still include them as part of the briefing team to the new leadership. Government technocrats often listen more to fellow government colleagues. Encourage them to do the briefing unless their presence will make matters worse.
Evidence-based advocacy
Advocacy messages based on evidence drawn from authorities respected by the decision-maker increase the possibility of an advocacy ask receiving a favorable outcome.
Framing of agenda
A family planning training package that encompasses all methods that pharmacists and pharmaceutical technologists have been allowed to provide enables an expanded choice for clients. It also allays concerns from authorities that the advocacy efforts are method driven. How you frame your advocacy agenda matters.
Know your decision-maker
Target the right decision-maker. Construct the right message and deliver it at the right time and in good company.
Next Steps
The training coordination committee established by MOH formed two subcommittees—the work plan and reporting sub-committees. As a next step, the work plan subcommittee will keep updating the implementation plan jointly with stakeholders to accelerate rolling out the trainings. The reporting subcommittee will liaise with the MOH Department of Monitoring Evaluation and Health Informatics to track family planning reporting by pharmacists and pharmaceutical technologists. AFP partner Jhpiego will continue to engage with key stakeholders and additional schools of pharmacy to advocate for introduction of injectable contraceptives in the pharmacy pre-service curriculum as well digitalization of the curriculum.
To learn more about Jhpiego’s work, visit www.jhpiego.org.
To learn more about Advance Family Planning’s work, visit www.advancefamilyplanning.org.





